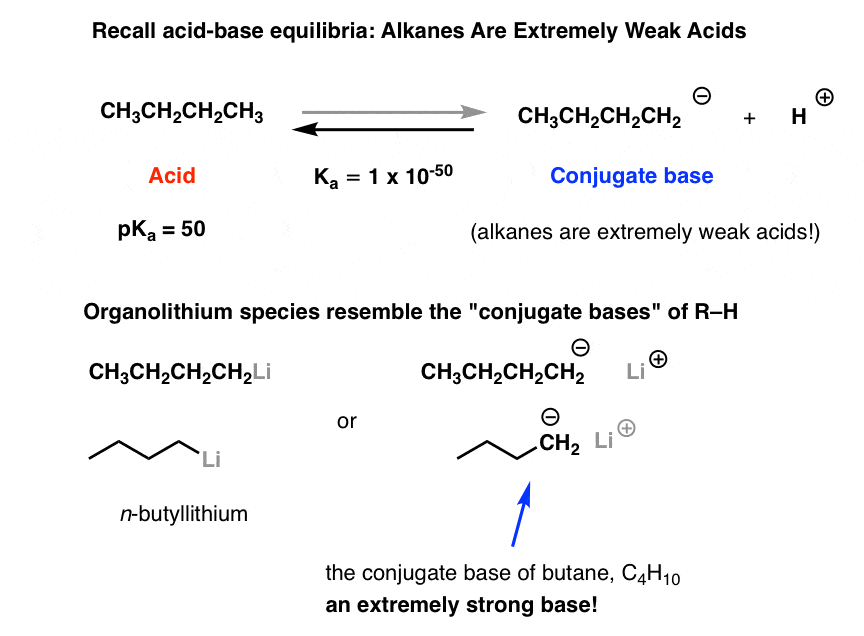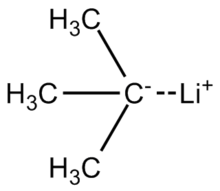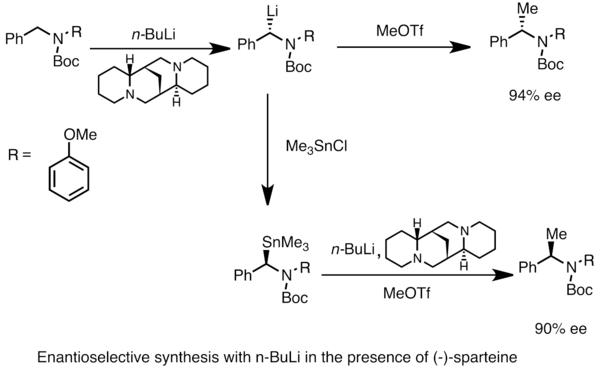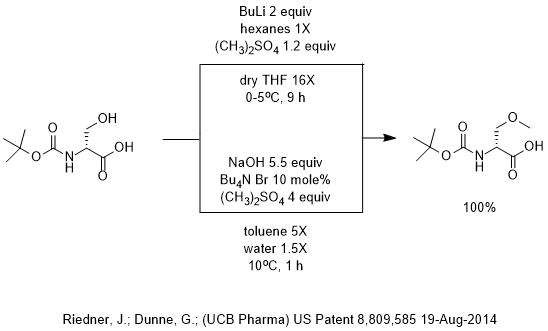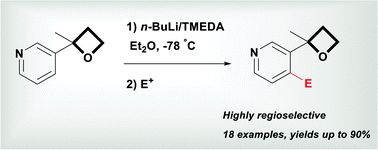
Highly regioselective lithiation of pyridines bearing an oxetane unit by n- butyllithium - Chemical Communications (RSC Publishing)

Acros Organics AC187540090 sec-Butyllithium, 1.3M solution in cyclohexane/hexane (92/8) (9g) from Cole-Parmer India

Ministry of Chemistry - n-#Butyllithium (abbreviated #nBuLi) is an organolithium reagent. It is widely used as a polymerization initiator in the production of elastomers such as #polybutadiene or styrene-butadiene-styrene (#SBS). Also, it

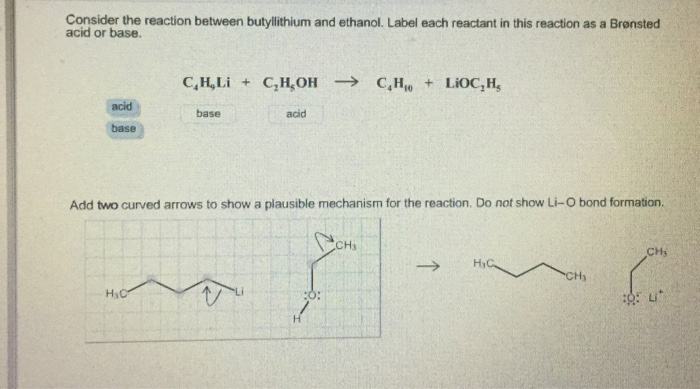
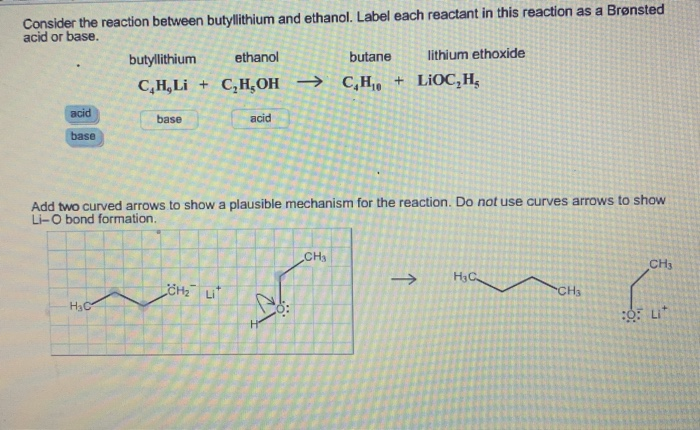
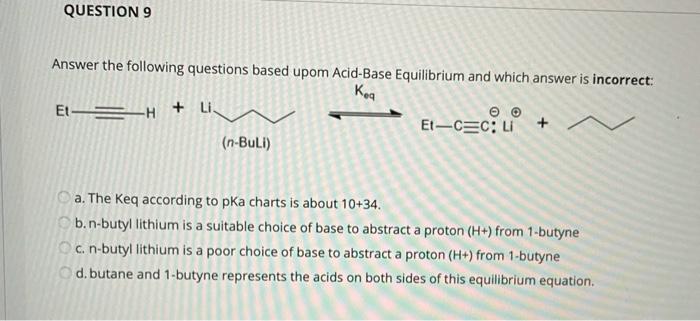
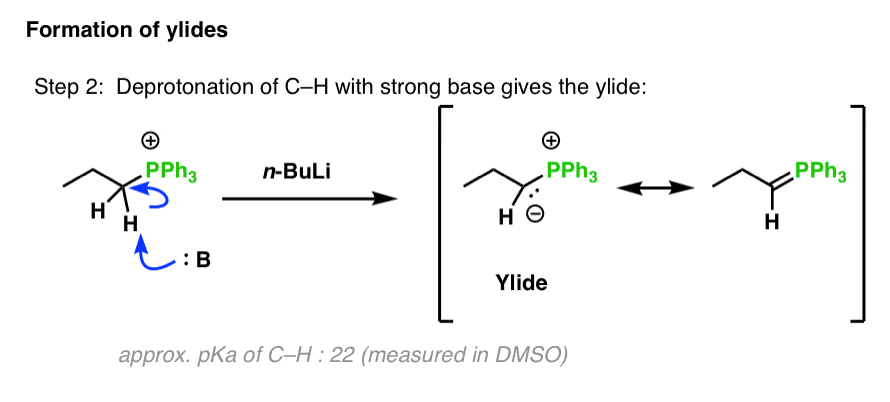
SOLVED: Match the species below to their appropriate, relative basic strength. butyllithium acetate alkoxide acetate Weakest Base alkoxide 2) Medium strength base 3) Strongest Base butyllithium

n-Butyllithium/N,N,N',N'-Tetramethylethylenediamine-Mediated Ortholithiations of Aryl Oxazolines: Substrate-Dependent Mechanisms | Journal of the American Chemical Society

Effect of Solvent on the Lithium−Bromine Exchange of Aryl Bromides: Reactions of n-Butyllithium and tert-Butyllithium with 1-Bromo-4-tert-butylbenzene at 0 °C | The Journal of Organic Chemistry